Resources
About Us
Medical Device Contract Manufacturing Market Size, Share, Forecast, & Trends Analysis by Device (Biochemistry, Immunoassay, CT, MRI, X-ray, Ultrasound, Pacemaker, Defibrillator, Oximeter) Services (Development, Manufacturing, QA) – Global Forecast to 2032
Report ID: MRHC - 1041138 Pages: 450 Sep-2024 Formats*: PDF Category: Healthcare Delivery: 24 to 72 Hours Download Free Sample ReportThe growth of this market is fueled by the increasing trend of outsourcing design, development, and manufacturing in the MedTech sector. Contract manufacturing offers various benefits, such as reduced development costs and time, flexibility to scale production according to demand fluctuations, and access to specialized manufacturing processes. Additionally, changing regulatory landscapes in the medical device industry compel companies to seek regulatory services and engineering expertise from contract manufacturers. Furthermore, the adoption of industry 4.0 technologies, coupled with rising healthcare expenditure and demand for quality healthcare in developing countries, presents growth opportunities for market players. Moreover, the cost advantages offered by contract manufacturers in emerging economies further contribute to market expansion.
The changing regulatory landscape in medical device contract manufacturing pertains to the dynamic rules and standards governing the production and distribution of medical devices by contract manufacturers. This encompasses updates in regulations concerning quality criteria, safety protocols, documentation requisites, and compliance procedures mandated by regulatory bodies like the FDA (Food and Drug Administration) in the U.S. or the EMA (European Medicines Agency) in Europe. Such alterations necessitate contract manufacturers to adapt their operations, technologies, and quality assurance systems to adhere to the revised regulatory mandates.
Contract manufacturers proactively stay abreast of regulatory revisions across manufacturing, technology, and quality management domains. Equipped with dedicated teams proficient in medical device regulatory affairs, they swiftly implement necessary adjustments in manufacturing and development practices in response to regulatory updates. This proactive approach alleviates the burden on medical device companies, sparing them from frequent modifications to manufacturing processes or technologies prompted by regulatory shifts. Consequently, medical device companies often favor the expertise and adaptability offered by contract manufacturers in navigating the evolving regulatory landscape.
Recent technological advancements have revolutionized the contract manufacturing process, ushering in heightened efficiency, enhanced customization capabilities, and improved communication channels between manufacturers and clients. These advancements empower contract manufacturers to expedite product delivery while upholding superior quality standards. In the realm of medical device contract manufacturing, companies have embraced various technological innovations to optimize their operations, including:
Advanced Automation Integration: Integration of AI and robotics to enhance efficiency.
Contract manufacturers gain a competitive edge in manufacturing processes through the active adoption of advanced technologies. Meanwhile, when medical device companies opt to manufacture products in-house, they often face significant investment requirements, limiting their capacity for other initiatives. Consequently, there is a growing inclination towards outsourcing among medical device companies.
Click here to: Get a Free Sample Copy of this report
Mergers & acquisitions in the medical device contract manufacturing industry help the companies strengthen their core capabilities, expand their services, and diversify their capabilities. M&A can also help companies reduce costs, shorten time to market, and improve return on investment. Additionally, it also helps the companies in:
Vertical integration to optimize supply chains
Following are some of the recent acquisitions:
In the highly competitive healthcare sector, medical device firms recognize the importance of concentrating on core strengths such as research and development (R&D), marketing, and sales to stay ahead. Outsourcing non-core functions like manufacturing to specialized contract manufacturers enables these firms to use resources more effectively, channeling them towards strategic endeavors like R&D and market expansion.
Contract manufacturers often possess specialized knowledge in manufacturing processes, materials, and technologies, providing valuable insights and capabilities to medical device firms. Additionally, outsourcing manufacturing enhances flexibility and scalability, allowing firms to adapt to fluctuations in demand and product lifecycles without significant capital investment. By prioritizing core competencies and outsourcing manufacturing, medical device companies can boost efficiency, flexibility, and innovation while reducing costs and mitigating risks in the healthcare industry.
Industry 4.0 is revolutionizing the medical device contract manufacturing market through the integration of digital technologies such as additive manufacturing, robotics, and the Internet of Things (IoT). These advancements empower contract manufacturers to create intricate medical devices with precision and adaptability, reducing time-to-market and facilitating customization. Digitalization and connectivity improve operational efficiency by enabling real-time monitoring and predictive maintenance, while smart manufacturing systems ensure product quality and compliance with regulations.
Technologies like blockchain enable end-to-end traceability and transparency, while Industry 4.0 facilitates mass customization and personalized manufacturing through digital design tools and virtual simulations. Supply chain optimization is achieved through digital supply chain platforms and predictive analytics, minimizing lead times and mitigating supply chain risks. Overall, Industry 4.0 presents medical device contract manufacturers with opportunities to innovate, streamline operations, and provide value-added services to their clients.
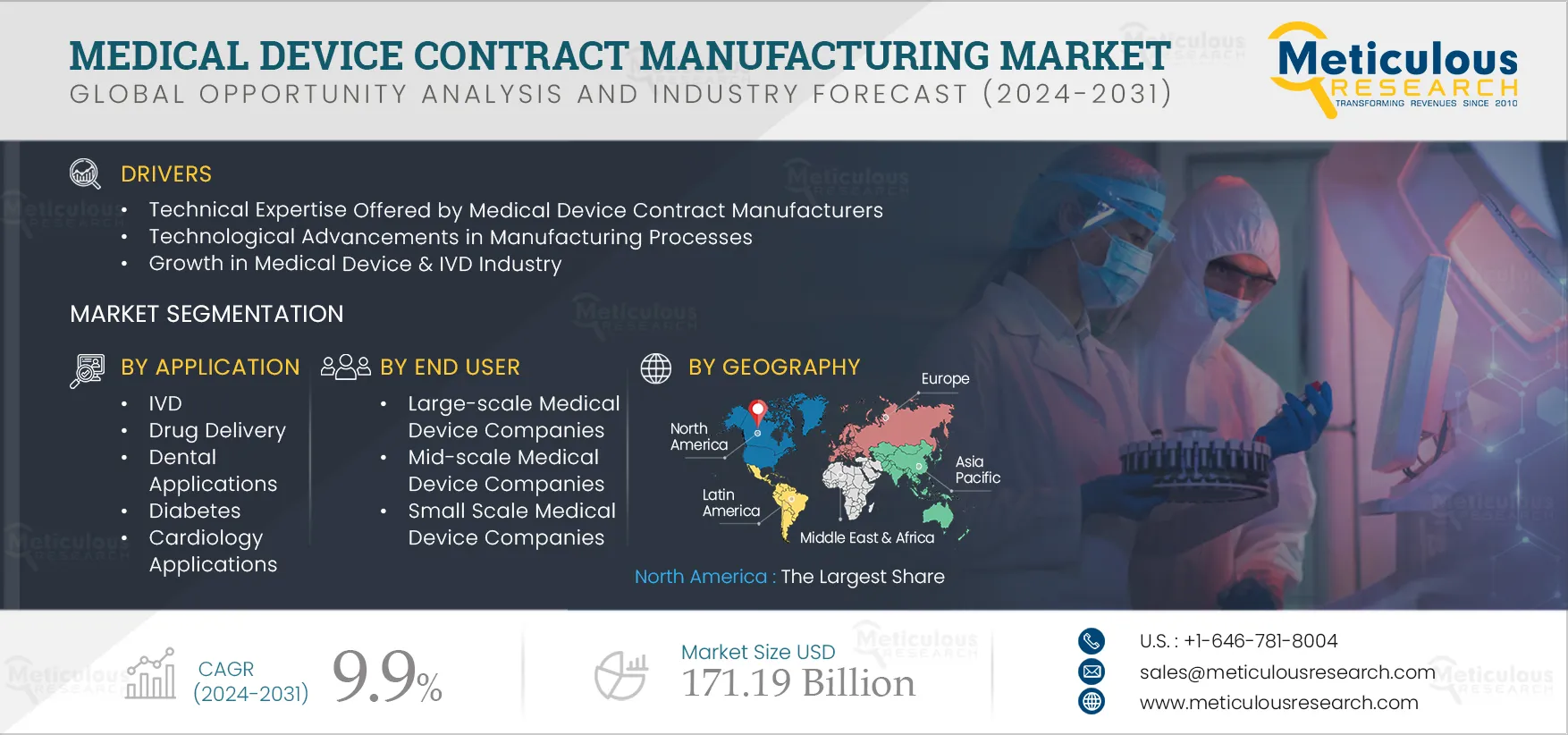
Based on device, the market is segmented into IVD devices, IVD consumables, imaging devices, cardiovascular devices, imaging devices, drug delivery devices, orthopedic devices, endoscopy devices, ophthalmology devices, dental devices, patient monitoring devices, anesthesia devices, mobility aids, surgical systems, consumables, and other devices. In 2025, the IVD devices segment is expected to account for the largest share of 21% of the global medical device contract manufacturing market.
The large market share of this segment is attributed to the high burden of chronic & infectious diseases propelling R&D in IVD. High preference for IVD contract manufacturing to IVD CDMOs. Within the IVD industry, there is a high preference for outsourcing of development and manufacturing of immunoassay analyzers due to the high demand for immunoassay technology contributes to the large segmental share. This high demand for immunoassay can be attributed to the benefits of immunoassay technologies such as rapid, convenient, and accurate results in the detection and quantitation of targets; the continuous development of new biomarkers; the cost-benefit of immunoassay technologies; and the growing adoption of automated platforms for ELISA.
However, the drug delivery devices segment is projected to witness the highest growth rate of 11.2% during the forecast period of 2025–2032. This growth is driven by rising chronic disease incidence, growing adoption of biologic drugs and specialty pharmaceuticals, growing emphasis on patient-centric care, and a trend of self-administration of medications.
Based on risk type, the global medical device contract manufacturing market is segmented into low-risk medical devices, moderate-risk medical devices, and high-risk medical devices. In 2025, the moderate-risk medical devices segment is expected to account for the largest share of the medical device contract manufacturing market. The moderate risk medical devices include catheters, blood pressure cuffs, pregnancy test kits, and blood transfusion kits, among others.
Outsourcing the development and manufacturing of moderate-risk medical devices is feasible with careful attention to regulatory compliance. However, for high-risk devices, concerns about intellectual property and maintaining control often limit outsourcing opportunities. Additionally, factors contributing to the significant share of the segment include the frequent utilization of devices like syringes, the increasing elderly population driving demand for catheters, and widespread adoption of self-pregnancy test kits. Owing to the rising adoption of medical devices falling under the moderate risk category due to the growing adoption of home healthcare and the rising geriatric population, the same segment is expected to register the highest CAGR during the forecast period of 2025-2032.
Based on services, the global medical device contract manufacturing market is segmented into design & development, manufacturing, testing and validation, quality assurance, implementation and maintenance services, packaging services, regulatory affairs services, and other services. In 2025, the design & development segment is expected to account for the largest share of 24% of the global medical device contract manufacturing market. The large market share of this segment is attributed to the growing demand for advanced medical devices and the high cost associated with the design and development of medical devices.
The process of designing and developing a medical device encompasses various expenses, including prototype development, component costs, regulatory compliance testing, clinical trials, and administrative expenses. The overall cost of medical device development varies based on factors such as device complexity and technological sophistication. High-risk devices, requiring extensive clinical trials and safety testing, often incur higher costs due to the expertise needed to ensure their safety and reliability.
The increasing demand for advanced medical devices to support diagnostics, monitoring, and treatment is driving the need for technical expertise in medical device development. Concurrently, there is a trend within the medical technology industry towards outsourcing the development and manufacturing of medical devices. This shift allows companies to leverage the specialized capabilities of contract manufacturers, streamlining the development process and enhancing the overall efficiency of bringing innovative medical devices to market. Owing to this, the same segment is expected to grow with the highest CAGR of 11.7% over the forecast period.
Based on application, the global medical device contract manufacturing market is segmented into IVD, drug delivery, dental applications, diabetes, diagnostic imaging applications, cardiology applications, orthopedic applications, endoscopy applications, surgical applications, and other applications. In 2025, the IVD segment is expected to account for the largest share of the market. This large market share is attributed to the growing demand for IVD tests due to the high burden of communicable and non-communicable diseases, the high adoption of predictive testing, and the growing preference for IVD companies to outsource the development and manufacturing of IVD devices.
However, the drug delivery segment is expected to grow at the highest CAGR during the forecast period of 2025-2032. The factors attributed to this growth are the growing adoption of self-administration of medications, increasing preference for home healthcare, innovations such as smart inhalers, and growing initiatives by contract manufacturers to strengthen their manufacturing capabilities of drug delivery devices.
Based on end user, the global medical device contract manufacturing market is segmented into large-scale medical device companies, mid-scale medical device companies, and small-scale medical device companies. In 2025, large-scale medical device companies are expected to account for the largest share of the market. Large-scale manufacturers benefit from economies of scale, reducing manufacturing costs related to workforce wages, production machinery, and infrastructure. Moreover, they often have the opportunity to diversify into other business segments. Large-scale medical device companies commonly leverage the services of contract manufacturers, particularly for manufacturing accessories and components.
On the other hand, the mid-scale medical device companies segment is poised to experience rapid growth during the forecast period 2025-2032. This growth is driven by the increasing focus of mid-sized industries on innovation and product introduction while adhering to cost constraints. As a result, these companies turn to contract manufacturers for development and manufacturing services. Mid-sized medical device companies typically compete with larger counterparts based on product cost and technological superiority. By partnering with contract manufacturers equipped with advanced manufacturing capabilities, mid-sized companies can offer high-quality products at competitive prices to their customers.
In 2025, North America is expected to account for the largest share of 44% of the global medical device contract manufacturing market. Among the U.S. and Canada, in 2025, the U.S. is expected to account for the largest share of the medical device contract manufacturing market in North America. The large market share is attributed to the well-developed manufacturing infrastructure, the presence of leading contract manufacturers in the U.S., the high adoption of advanced manufacturing technologies, and the high demand for medical devices. Additionally, the academia and industry collaboration in the country leads to technological manufacturing capabilities, further contributing to the large share.
However, Asia-Pacific is slated to register the highest CAGR of 11.9% during the forecast period. The growth of this region is attributed to the availability of skilled labor, rising quality standards in manufacturing as well as finished medical devices, government initiatives for local production of medical devices, and good accessibility and cheaper raw materials for medical devices in the region.
The cost-effectiveness of labor in Asia stands out as a key factor. Labor expenses in the Asia-Pacific region are not only economical but also come with a high level of skill. Additionally, other overhead costs such as rent, utilities, and raw materials tend to be significantly lower in Asia-Pacific compared to the United States and European countries. While China is renowned for its cost-efficient manufacturing, countries like Vietnam and India have emerged as competitive destinations for medical device production, particularly for standard devices.
The report offers a competitive analysis based on an extensive assessment of the leading players’ product portfolios, geographic presence, and key growth strategies adopted in the last 3–4 years. The key players operating in the global medical device contract manufacturing market are Jabil Inc. (U.S.), Gerresheimer AG (Germany), Nordson Corporation (U.S.), Teleflex Incorporated (U.S.), Celestica Inc. (Canada), Plexus Corp (U.S.), SteriPack Group Ltd (U.S.), Nortech Systems, Inc. (U.S.), Invetech Pty. Ltd. (Australia), Flextronics International, LTD. (Singapore), Benchmark Electronics Inc. (U.S.), Integer Holdings Corporation (U.S.), STRATEC SE (Germany).
In 2025, Fujirebio Holdings, Inc. (Japan) and Agappe Diagnostics Ltd (India) today announced that they have entered into an agreement of Contract Development and Manufacturing Organization (CDMO) partnership for Cartridge based CLIA system reagents manufacturing project for the immunology equipment Mispa i60 and Mispa i121. The analyzers and reagents will be sold under Agappe’s brand.
In 2024, Sysmex Corporation (Japan) entered into an agreement with Fujirebio Holdings, Inc. (Japan), to expand their Contract Development and Manufacturing Organization (CDMO) partnership for Sysmex's Automated Immunoassay System HISCL.
|
Particular |
Details |
|
Page No |
450 |
|
Format |
|
|
Forecast Period |
2025-2032 |
|
Base Year |
2024 |
|
CAGR |
9.9% |
|
Market Size (Value) |
$171.19 billion by 2032 |
|
Segments Covered |
By Devices
By Services
By Risk Type
By Application
By End User
|
|
Countries Covered |
North America (U.S., Canada), Europe (Germany, France, U.K., Spain, Italy, Sweden, Switzerland, Belgium, Rest of Europe), Asia-Pacific (China, Japan, India, Singapore, Rest of APAC), Latin America (Brazil, Mexico, Rest of Latin America), and Middle East & Africa (South Africa, Saudi Arabia, Rest of Middle East & Africa) |
|
Key Companies |
Jabil Inc. (U.S.), Gerresheimer AG (Germany), Nordson Corporation (U.S.), Teleflex Incorporated (U.S.), Celestica Inc. (Canada), Plexus Corp (U.S.), SteriPack Group Ltd (U.S.), Nortech Systems, Inc. (U.S.), Invetech Pty. Ltd. (Australia), Flextronics International, LTD. (Singapore), Benchmark Electronics Inc. (U.S.), Integer Holdings Corporation (U.S.), STRATEC SE (Germany). |
The medical device contract manufacturing market covers the market sizes & forecasts of outsourcing high, low, and moderate-risk medical device manufacturing. The medical device contract manufacturing market studied in this report involves the value analysis of various segments and sub-segments of the medical device contract manufacturing market at regional and country levels.
The global medical device contract manufacturing market is projected to reach $171.19 billion by 2032, at a CAGR of 9.9% during the forecast period.
In 2025, the design & development segment is expected to account for the largest share of the medical device contract manufacturing market.
The medical device contract manufacturing market is driven by the growing preference for outsourcing of development & manufacturing of medical devices, technological advancements in manufacturing processes, and the growth in the medical device and IVD industry. Moreover, Industry 4.0 and growing healthcare expenditure & demand for quality healthcare are expected to generate growth opportunities for players operating in the medical device contract manufacturing market.
The key players operating in the global medical device contract manufacturing market are Jabil Inc. (U.S.), Gerresheimer AG (Germany), Nordson Corporation (U.S.), Teleflex Incorporated (U.S.), Celestica Inc. (Canada), Plexus Corp (U.S.), SteriPack Group Ltd (U.S.), Nortech Systems, Inc. (U.S.), Invetech Pty. Ltd. (Australia), Flextronics International, LTD. (Singapore), Benchmark Electronics Inc. (U.S.), Integer Holdings Corporation (U.S.), STRATEC SE (Germany).
The countries India, China, and Singapore are projected to offer significant growth opportunities for the vendors in this market due to factors such as the availability of affordable workforce, growing adoption of advanced manufacturing technologies, government initiatives to promote medical device manufacturing, and availability of raw materials at a lower cost.
























Published Date: Jan-2025
Published Date: Nov-2024
Published Date: Nov-2024
Please enter your corporate email id here to view sample report.
Subscribe to get the latest industry updates